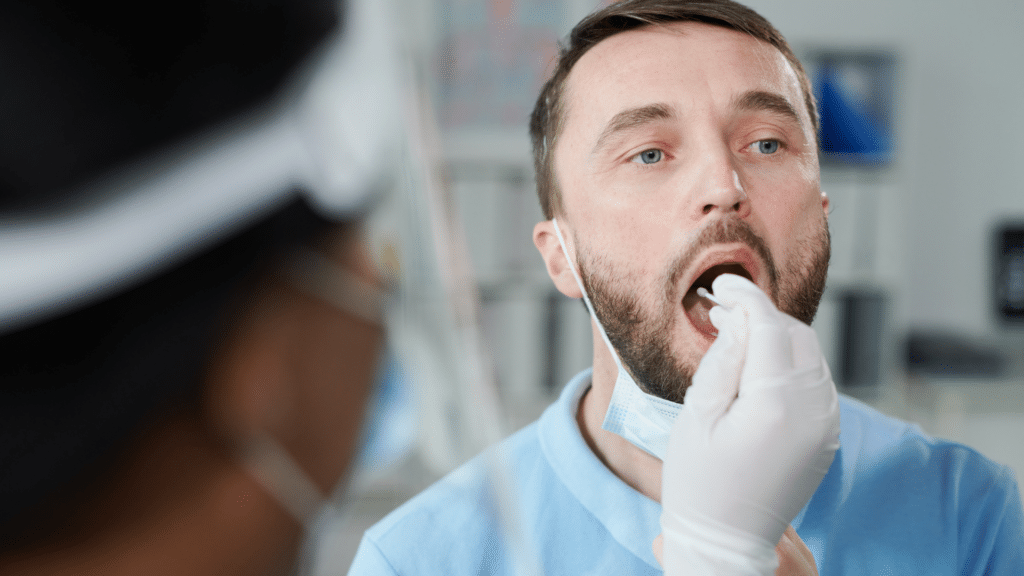
On May 2nd, 2023, multiple departments under the Department of Transportation (DOT) released a final ruling on procedures for drug and alcohol testing programs in the transportation workplace. This new procedure will include Oral Fluid Specimen Testing for Drugs. (other departments include; Federal Aviation Administration (FAA), Federal Railroad Administration (FRA), Federal Motor Carrier Safety Administration (FMCSA), and Federal Transit Administration (FTA))
By removing references to urine and adding references to oral fluid, this change is expected to provide employers with more options to conduct drug tests, thereby making it easier to achieve workplace safety goals. Despite the change, the use of urine is still acceptable, and employers have the option to choose between the two methods of testing.
The new regulation is set to take effect 30 days from the most recent update which was May 2nd, 2023, and DOT employers and industry professionals are encouraged to read the final ruling in preparation for implementation. The regulation provides detailed responses to comments received in response to the Notice of Proposed Rulemaking (NPRM), which can be valuable for those who have questions about the new regulations. Furthermore, terminology and requirements throughout 49 CFR Part 40 have been updated.
DATIA, an organization that supports the drug and alcohol testing industry, will continue to communicate, educate, and train its members to support compliance with these changes. It is important for employers to stay up-to-date with these changes and remain compliant to ensure the safety of their employees and the public.
Click HERE to view the complete DOT ruling.
What This Means For YOU
Even though there are now mandatory guidelines for drug testing using oral fluid in the transportation workplace, there are currently no laboratories certified by the U.S. Department of Health and Human Services (HHS) to conduct them. At least two laboratories will need to be HHS certified for oral fluid testing.
This requirement is mandated by the Department of Transportation (DOT), which specifies that the drug testing process must involve two separate HHS-certified labs. One lab would conduct screening and confirmation drug testing on the primary specimen, while the other lab would conduct split specimen drug testing on a second specimen.
The goal of this ruling is to prevent urine test “cheating” and offer an option when urine samples are insufficient for testing due to “shy bladder.” Additionally, oral fluid testing is also less intrusive than urine testing and it’s harder to “cheat” because the collection of oral fluid is directly observed.
It’s important to note that the detection window for urine drug testing is potentially longer versus the oral fluid drug testing method. Overall the addition of oral fluid drug testing in the transportation marketplace will mean that it’s harder to “cheat” on a drug test, thus contributing to achieving safety in the respective workplace environments. Additionally, this could also mean an increased demand for oral fluid testing on forensic laboratories.
Do you think your lab will need to make any changes due the new ruling? Let us know your thoughts in the comments below.
